02 Jul 2018
Focussed ultrasound used to improve effects of cancer drugs
Researchers have made a breakthrough in more precisely targeting drugs to cancers
Using ultrasound and lipid drug carriers (liposomes), a multi-disciplinary team of biomedical engineers, oncologists, radiologists and anaesthetists at the University of Oxford have developed a new way to improve the targeting of cancer drugs to tumours.
The new technology has been used in humans for the very first time, with ultrasound remotely triggering and enhancing the delivery of a cancer drug to the tumour.
"Reaching therapeutic levels of cancer drugs within a tumour, while avoiding side effects for the rest of the body is a challenge for all cancer drugs, including small molecules, antibodies and viruses," said Professor Constantin Coussios, Director of the Oxford Centre for Drug Delivery Devices (OxCD3) and of the Institute of Biomedical Engineering. "Our study is the first to trial this new technique in humans, and finds that it is possible to safely trigger and target the delivery of chemotherapy deep within the body from outside the body using focussed ultrasound. Once inside the tumour, the drug is released from the carrier, supplying a higher dose of chemotherapy directly to the tumour, which may help to treat tumours more effectively for the same or a lower systemic dose of the drug."
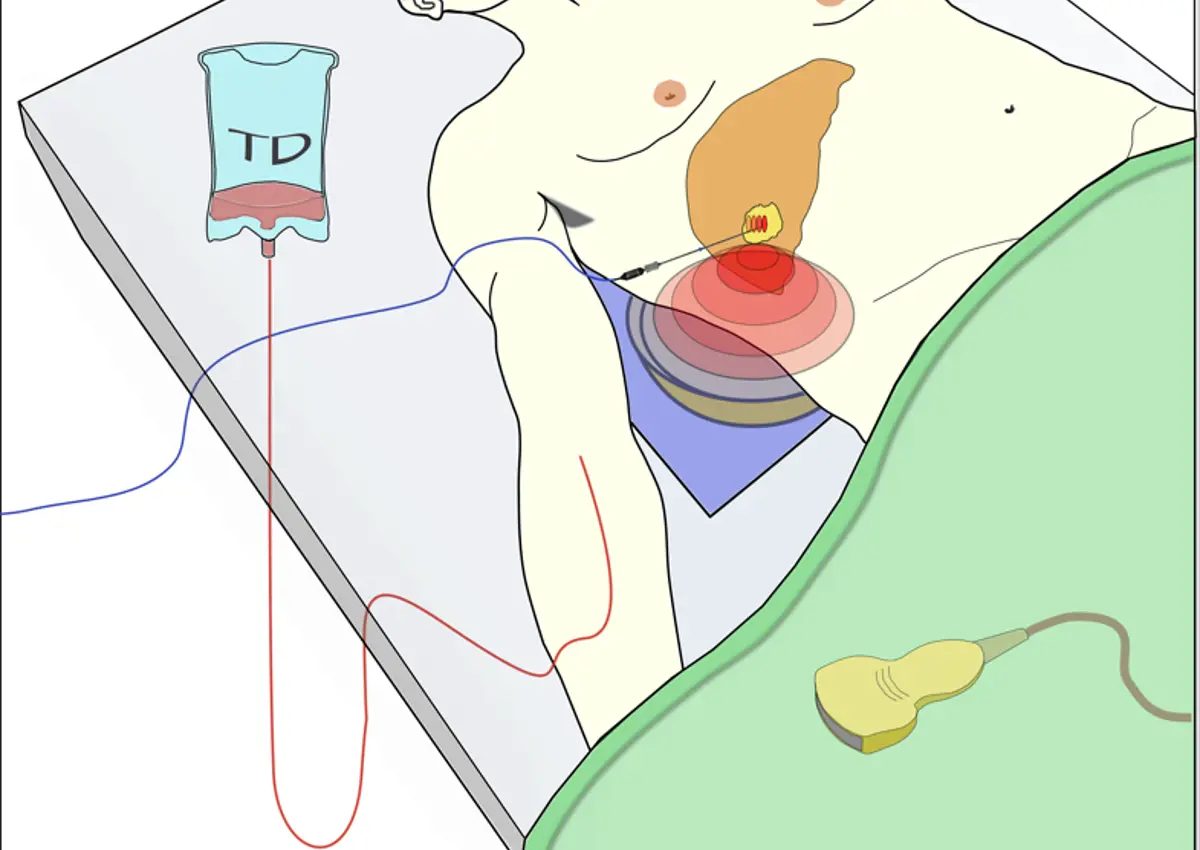
Published in The Lancet Oncology journal, the 10-patient phase 1 clinical trial used focussed ultrasound from outside the body to selectively heat liver tumours and trigger drug release from heat-sensitive carriers, known as thermosensitive liposomes. Building on over a decade of preclinical studies, the study demonstrated the ultrasound technique to be feasible, safe, and capable of increasing drug delivery to the tumour between two-fold and ten-fold in the majority of patients. Ongoing research worldwide is investigating the applicability of this technique to other tumour types, and future research could explore the combination of ultrasound with other drugs.
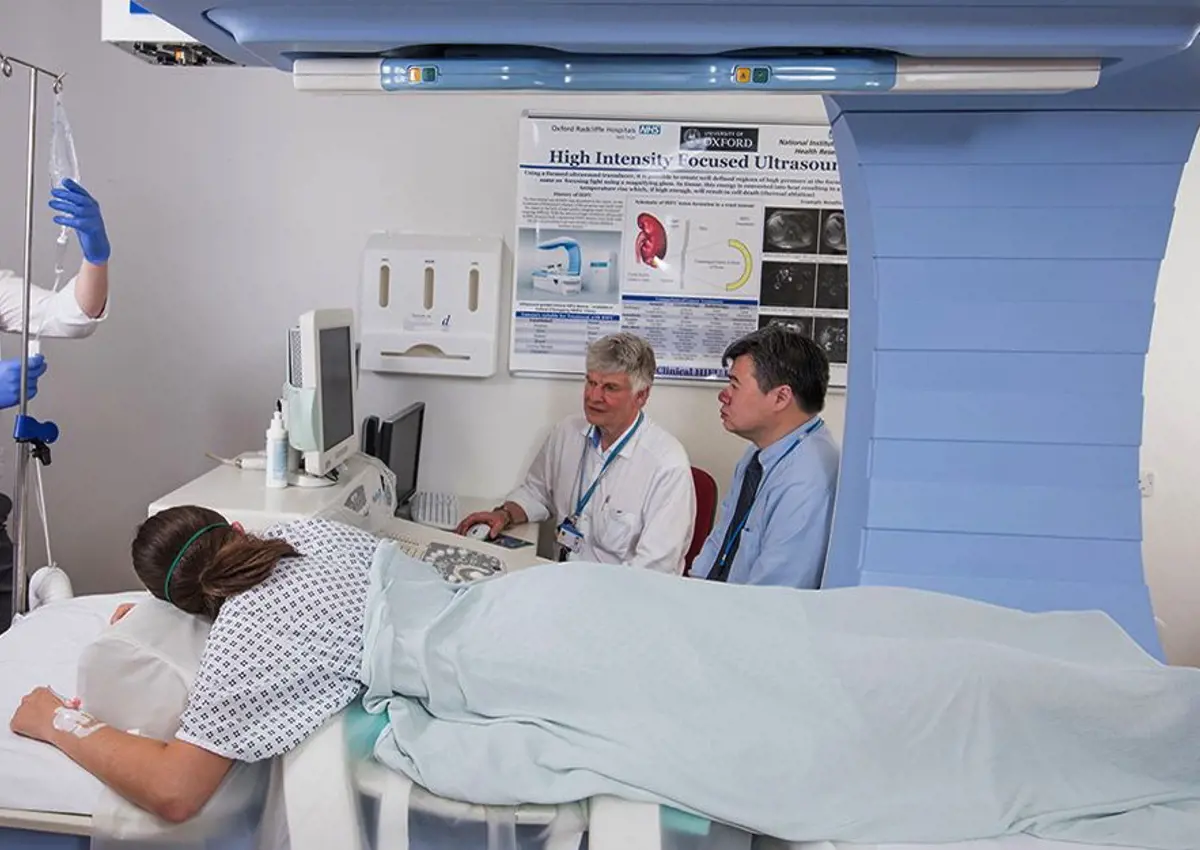
All 10 patients treated had inoperable primary or secondary tumours in the liver and had previously received chemotherapy. The procedure was carried out under general anaesthesia and patients received a single intravenous dose of 50 mg/m2 of doxorubicin encapsulated within low-temperature-sensitive liposomes (ThermoDox®, Celsion Corporation, USA). The target tumour was selectively heated to over 39.5oC using an approved ultrasound-guided focussed ultrasound device (JC200, Chongqing HAIFU, China) at the Early Phase Clinical Trials Unit at the Churchill Hospital in Oxford. In six out of 10 patients, the temperature at the target tumour was monitored using a temporarily implanted probe, whilst in the remaining four patients ultrasonic heating was carried out non-invasively.
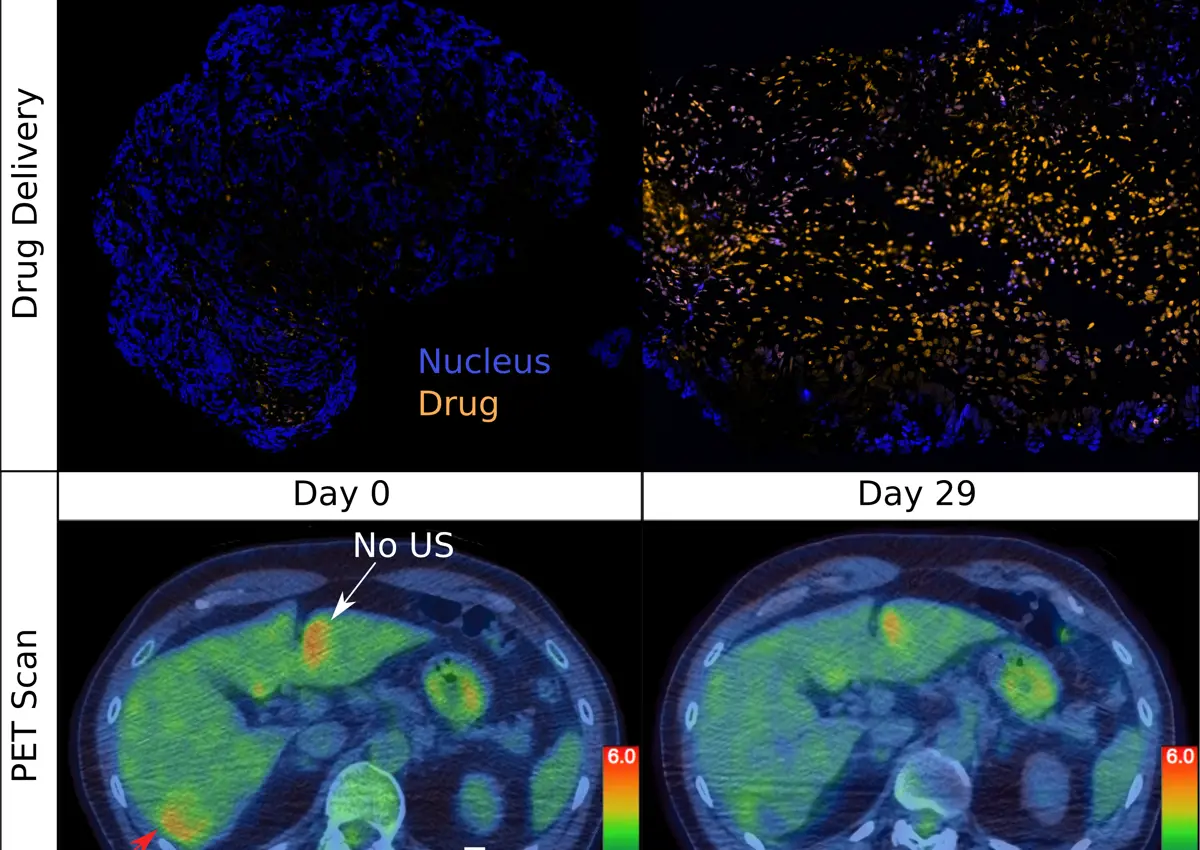
Before ultrasound exposure, the amount of drug reaching the tumour passively was low and estimated to be below therapeutic levels. In seven out of 10 patients, chemotherapy concentrations within the liver tumour following focussed ultrasound were between two and ten times higher, with an average increase of 3.7 times across all patients.
"Only low levels of chemotherapy entered the tumour passively. The combined thermal and mechanical effects of ultrasound not only significantly enhanced the amount of doxorubicin that enters the tumour, but also greatly improved its distribution, enabling increased intercalation of the drug with the DNA of cancer cells," said Dr Paul Lyon, lead author of the study.
"This trial offers strong evidence of the rapidly evolving role of radiology in not only diagnosing disease but also in planning, guiding and monitoring therapy. The treatment was delivered under ultrasound guidance and patients were subsequently followed up by CT, MRI and PET-CT, evidencing local changes in tumours exposed to focussed ultrasound," commented Professor Fergus Gleeson, radiology lead co-investigator for the trial.
"A key finding of the trial is that the tumour response to the same drug was different in regions treated with ultrasound compared to those treated without, including in tumours that do not conventionally respond to doxorubicin," added Professor Mark Middleton, principal investigator of the study. "The ability of ultrasound to increase the dose and distribution of drug within those regions raises the possibility of eliciting a response in several difficult-to-treat solid tumours. This opens the way not only to making more of current drugs but also targeting new agents where they need to be most effective. We can now begin to realize the promise of precision cancer medicine."
Read the full paper in The Lancet Oncology:Safety and feasibility of ultrasound-triggered targeted drug delivery of doxorubicin from thermosensitive liposomes in liver tumours (TARDOX): a single-centre, open-label, phase 1 trial